Plant bodies are shaped—literally—by their relationship with water. Each plant cell, with the exception of a few specialized cell types, is packed so full of water that pressures inside can reach 10atm, or 4-5x times the pressure in a car tire. Biologists refer to this pressure as turgor, and it's responsible for giving plants their firmness and structural integrity. But water also shapes plants in more subtle ways ..
Water availability is highly uneven on Earth's surface. Plants can live for many years, sometimes centuries, in a single place, while coastlines, rivers, and aquifers around them shift. Even in soils that appear well-watered, water molecules are heterogenously distributed. This means plant roots—the organs tasked with water uptake—have to deal with a unique challenge: capturing a dynamic resource from a fixed position.
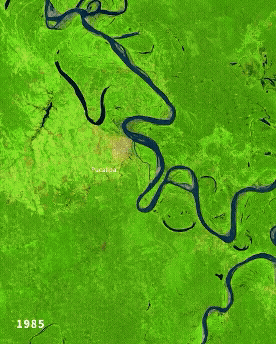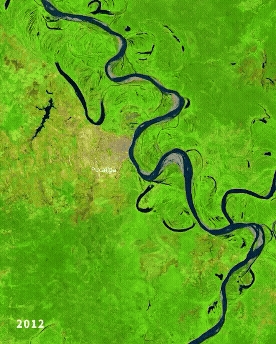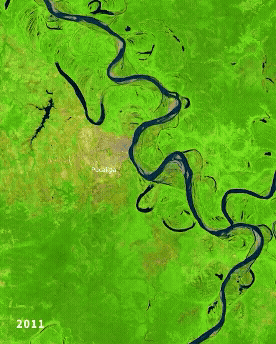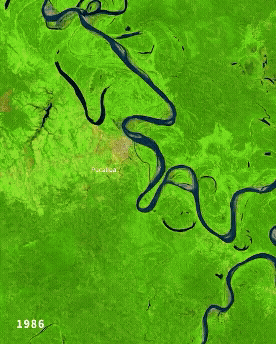

Left: Ucayali (Peru) river morphodynamics, by Cristian Yarasca. Right: Arabidopsis thaliana root system from Kutschera, L., Wurzelatlas, 1960.
The solution to this challenge is an exquisite sensitivity to moisture. Plants continuously monitor their surroundings and are capable of making developmental decisions in response to signs of water. For example, we've known for over a century that root tips can bend in the direction of water sources, a behavior dubbed hydrotropism that was first characterized by Julius Sachs in 1872 and by Charles and Francis Darwin a few years later.
The other known developmental responses—hydropatterning and xerobranching—are more subtle and weren't formally described until the 2010s by Bao et al in the Dinneny lab (wooo!) and Orman-Ligeza et al in in Europe. The discovery of hydropatterning and xerobranching reveal that root branching is regulated by water. In other words, plants use information about their environment to position and develop their new organs.
How plants achieve this feat of self-engineering is currently unknown. In a review written for BMC Biology, my colleagues and I summarize the most pertinent hypotheses. Here's a SparkNotes version.
Hypothesis 1: mechanical forces are generated as water moves in, out, and around cells. For example, tensile forces may build up or decrease in the plasma-membrane as cells experience different hydration states. A growing number of ion channels and other proteins responsive to mechanical forces has come to light in recent years. However, none have so far been convincingly found to regulate the root bending or branching responses to water described above. In some animals, mechanosensitive ion channels are required for perception but the search remains ongoing in plants.
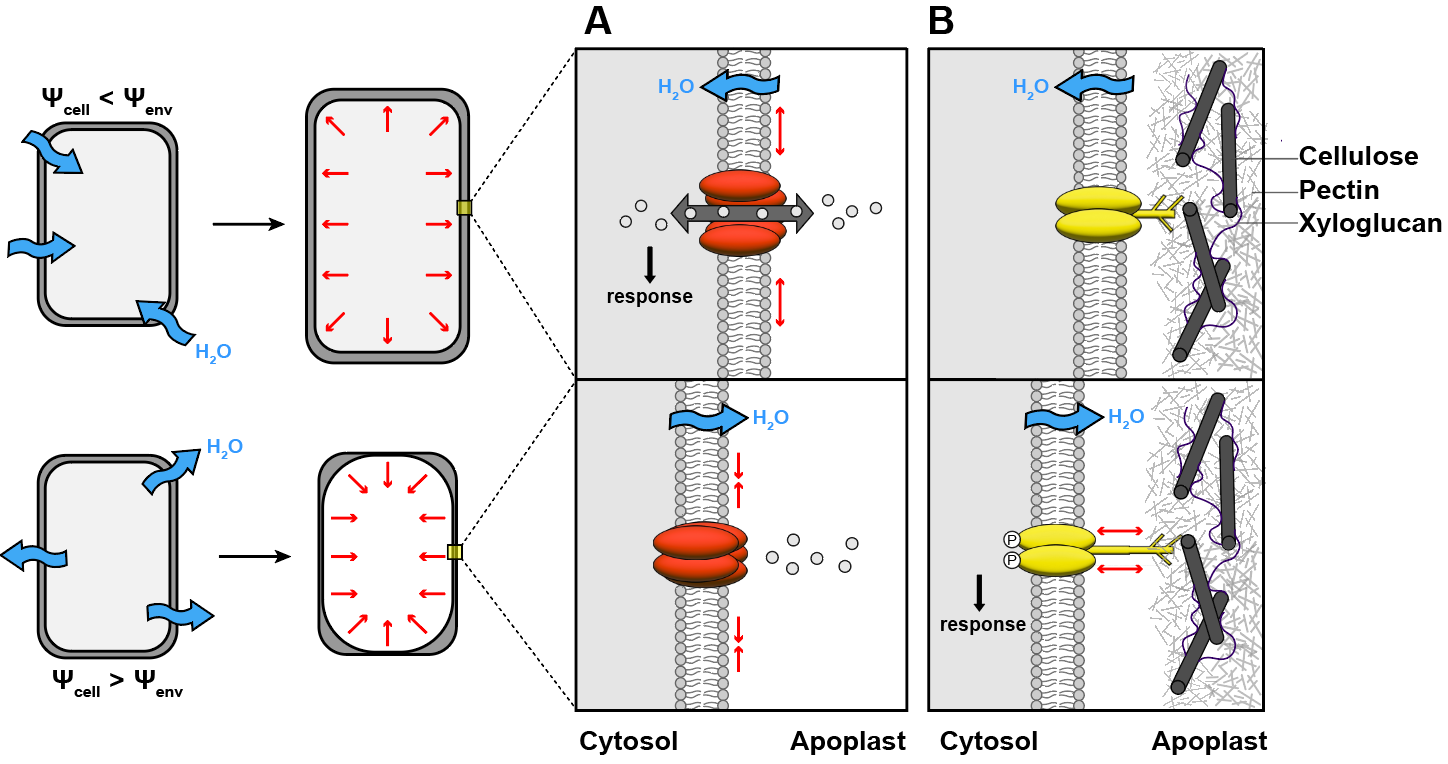
Possible mechanosensors for perception of water availability in plant cells.A) Some ion channels can be activated by tension in the plasma membrane. B) Membrane proteins with extracellular domains may respond to tension during separation of the membrane from the wall.
Hypothesis 2: sense-by-growth, whereby water's spatial distribution around a root is translated into a state of disequilibrium inside the root. Sounds confusing I know. But remember that plant cells grow by uptaking water from the environment. Now imagine that cells on one side of a root have easy access to moisture, while cells on the other have a harder time 'finding' water. As a result of this uneven access to water, cells in the root experience different hydration states, at least transiently in the period of time during which the root is growing. This idea was first suggested by Neil Robbins in his PhD thesis but remains to be tested experimentally. This is where my work comes in ... See my osmosensor project for more detail on this.
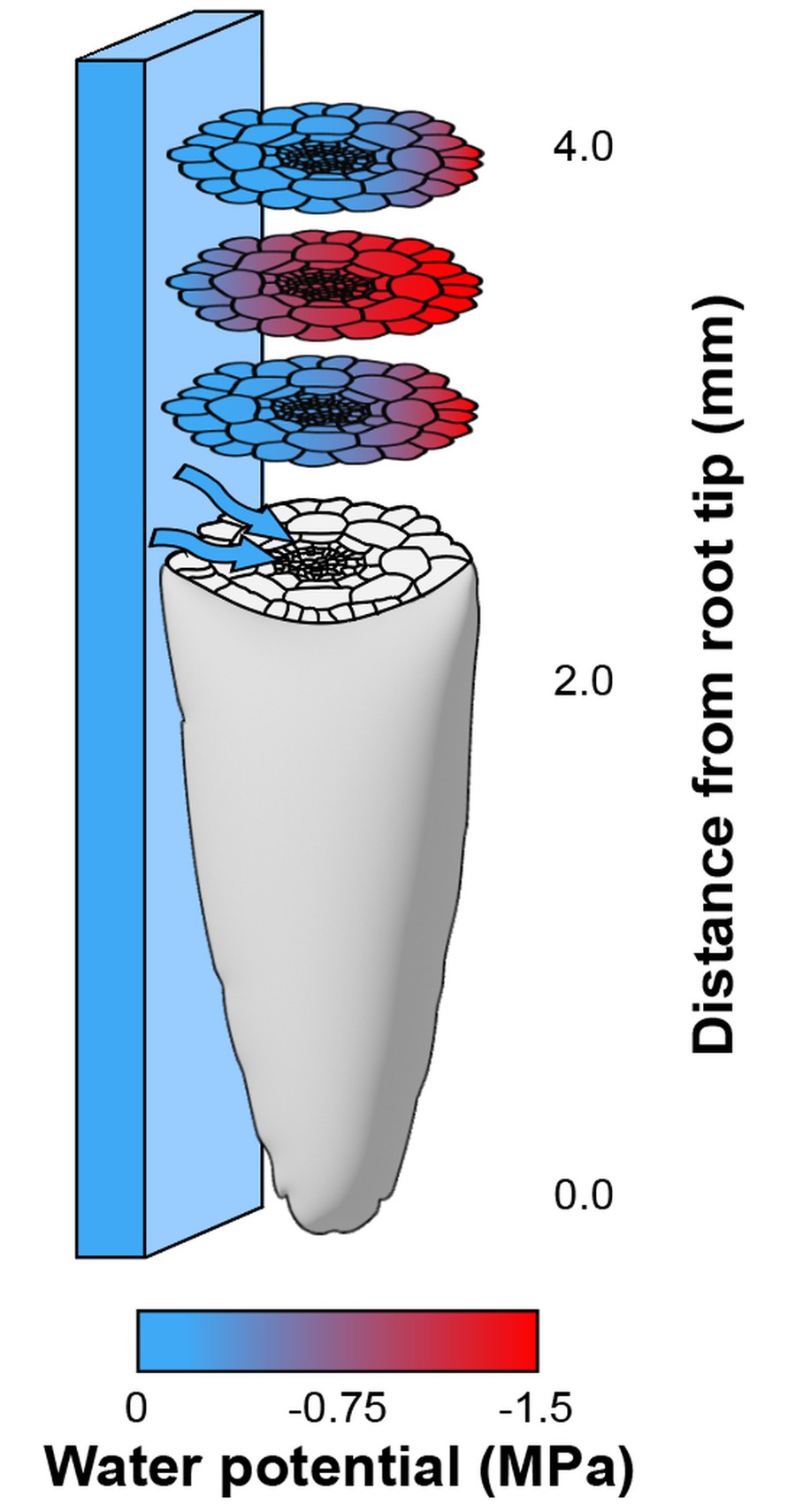
Computational model of the sense-by-growth hypothesis. In the growth zone, represented here 2-4mm from the root tip, an internal water potential gradient is formed from uneven access to water.
Hypothesis 3: ethylene signaling is a canonical hormone pathway that regulates many processes from fruit ripening (brown bananas!) to organ development. Ethylene and ethylene-associated genes were recently shown to control hydropatterning, specifically by inhibiting lateral root formation on the air-side of roots. How ethylene achieves this side-specifity is unknown. Drawing from known information about the ethylene biosynthetic pathway and gas diffusion dynamics, we propose that the hormone may be preferentially synthesized at air-interfaces where it achieves its inhibitory effect locally before diffusing away. A similar mechanism has been proposed to underpin the root's ability to sense soil compaction.
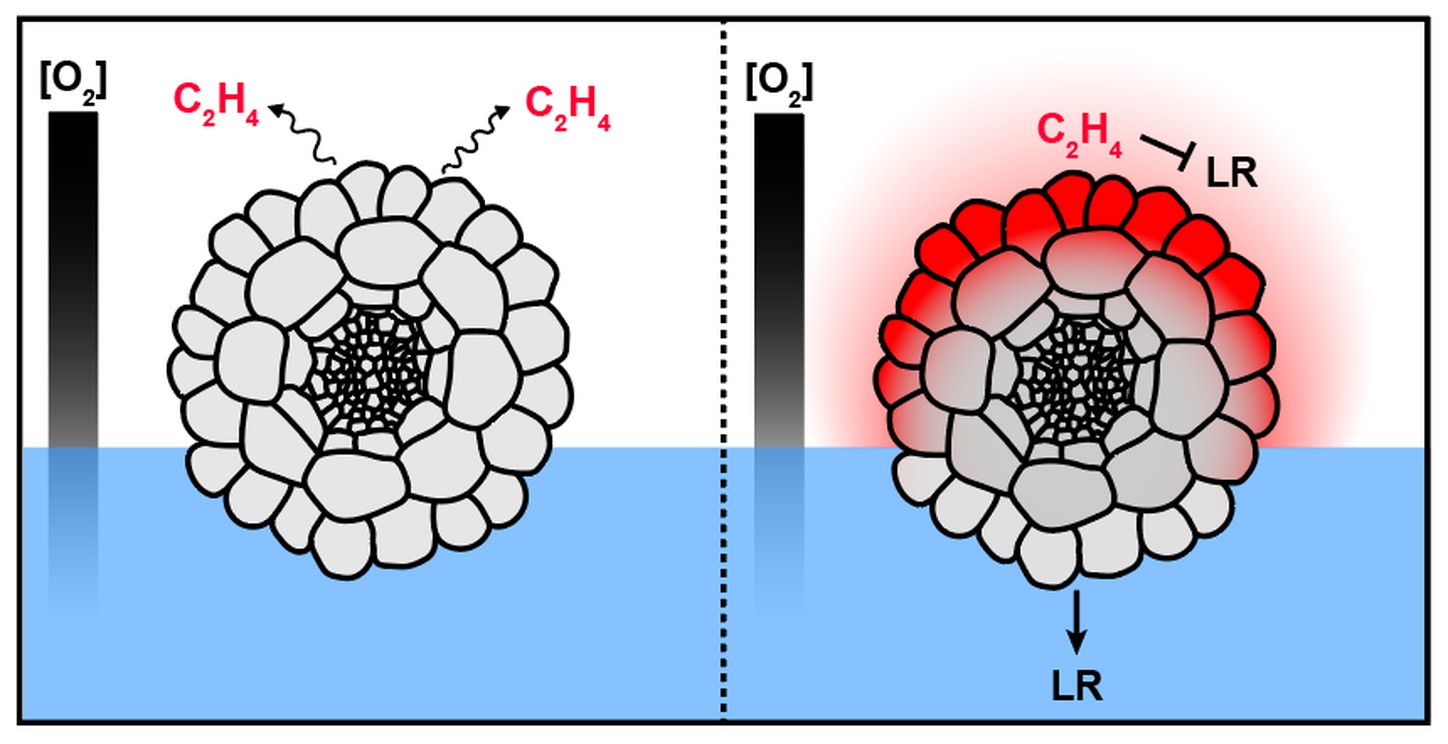
Theoretical model of air-side lateral root inhibition by ethylene. In this model, roots don't perceive water directly but rather suppress organ development by chemically detecting the presence of air.