Plant cells are encased inside a rigid polysaccharide cell-wall that grants them structural strength and protects them from the environment. The integrity of this sugary matrix is continuously monitored to respond to external stresses and selectively weakened during growth to accomodate cell elongation.
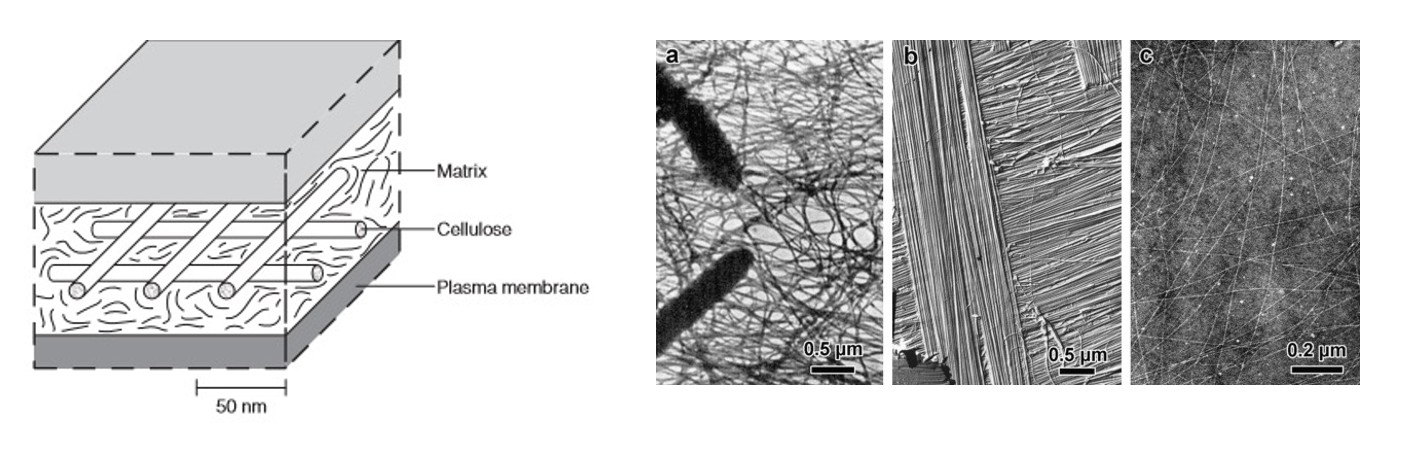
Left: macrostructure of the cell boundary from BrainKart. Right: bacterial, algal and plant cellulose microfibril structure reprinted in Ogawa, 2019 with permission from the original authors.
The interface between cell walls and membranes is a mysterious, fast-changing world. Under non-stressed conditions, the cell membrane is tightly appressed against the wall from the pressure of turgor. Under some stress conditions—for instance hyperosmotic shock, which causes rapid cell water loss—the membrane can detach from the wall. Forces generated during this transition are believed to represent a source of signal perceived by stretch-activated ion channels and mechanically sensitive proteins. Previous observations by the botanist Karl Hecht in 1912 revealed that membranes do not completely detach from the cell wall but maintain attachment points called Hechtian strands. The nature of these attachment points remains elusive, but they are thought to be important components of sensing and stress-response signaling.
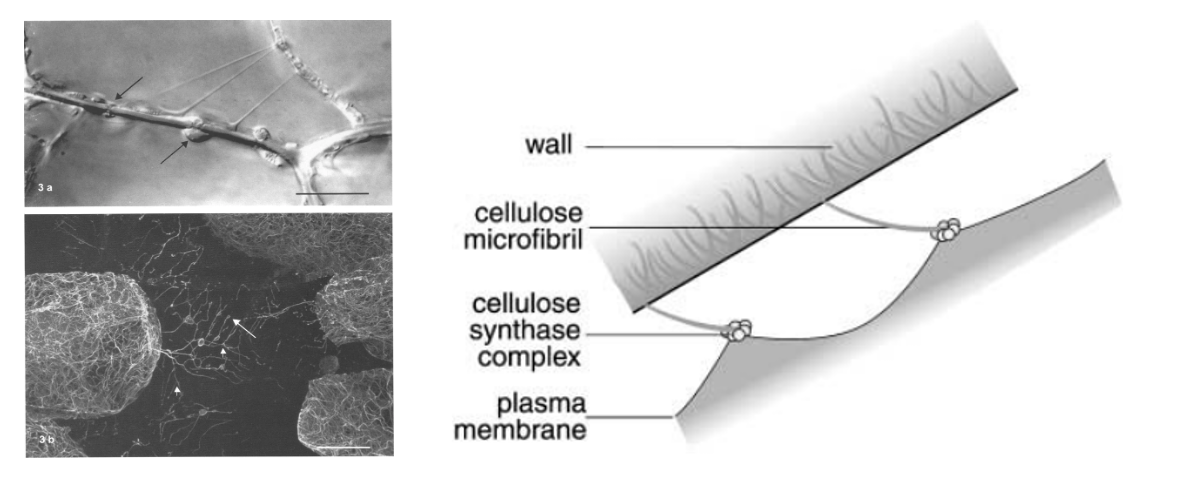
Left: differential interference contrast (DIC) microscopy of Hechtian strands in Allium cepa (onion) epidermal cells from Lang-Pauluzzi and Gunning, 2000. In panel b, microtubules are stained to reveal cytoskeletal elements within the strands. Right: "Velcro hypothesis" scheme proposed by Lang, 2003, whereby nascent cellulose-microfibrils link the cell-wall to the plasma-membrane where cellulose synthase complexes reside.
All work described below is a collaboration between Dr. Yue Rui and the Dahlberg Lab at Stanfords Linear Accelerator Center (SLAC). These images could not have been collected without the incredible patience of Magdi Zaoralova and the guidance of Peter Dahlberg. Using a modified sample preparation workflow and super-resolution TEM imaging, we were able to resolve subcellular features directly in Arabidopsis root tissue. To our knowledge, these data represent the first cryoprotectant-free electron tomograms of live plant cells in their native context. These are snapshots of real biology captured at near-atomic scales.


Left: tomographic reconstruction of a TEM tilt-series. Right: segmentation of subcellular features in Dragonfly. Segmented features: ribosomes, cellulose microfibrils, membranes, microtubules.
To investigate the effects of osmotic shock on the wall-membrane interface, we treated 5-day old Arabidopsis seedlings with 1M sorbitol and immediately froze samples for downstream imaging. In this experiment, we successfully resolved wall-membrane features of hyperosmotically shocked root cells and revealed the presence of membrane topologies resembling Hechtian strands.
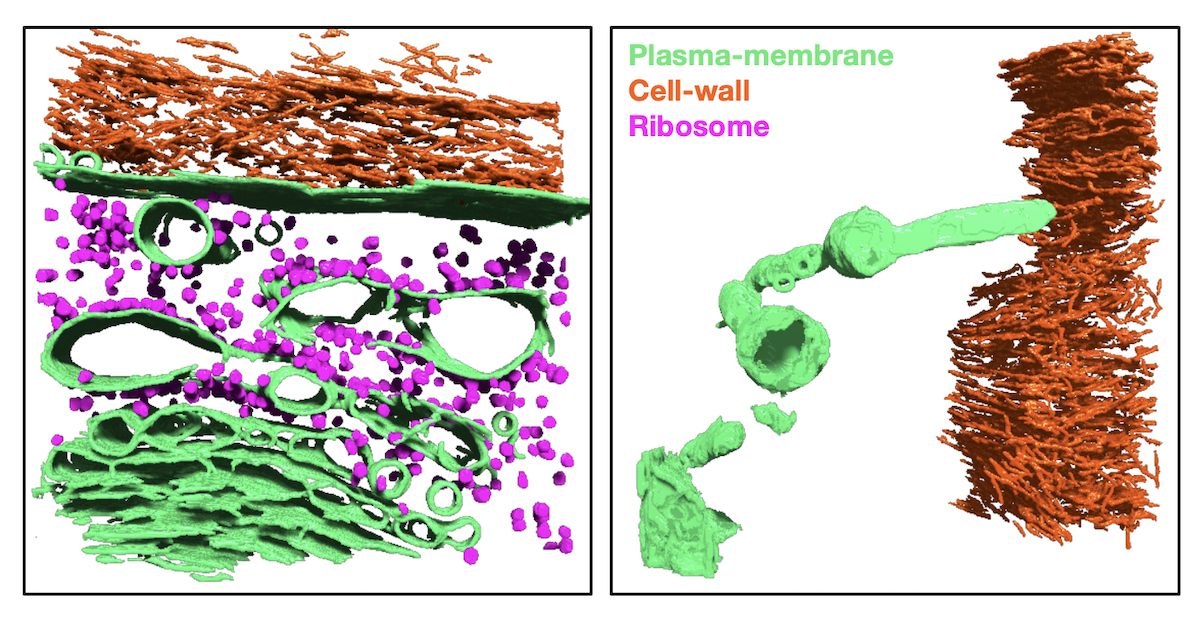
Dragonfly segmentations of cryo-ET data collected by Magdi Zaorolova. Left: wall-membrane interface in unstressed Arabidopsis root. Right: wall-membrane interface of Arabiodpsis root treated with 1M sorbitol. Finger-like plasma-membrane structure resembling a Hechtian strand is visible.
This work is ongoing and will be partially published in an upcoming manuscript. A separate methods paper describing sample-preparation is also in the works.